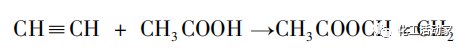Vinyl acetate (Vac), nke a makwaara dị ka vinyl acetate ma ọ bụ vinyl acetate, bụ mmiri mmiri na-enweghị atụ na-enweghị atụ na okpomọkụ na nrụgide nkịtị, yana usoro ihe omimi nke C4H6O2 na ihe dị arọ nke 86.9. Vac, dị ka otu n'ime ihe ndị a na-ejikarị eme ihe n'ụwa, nwere ike ịmepụta ihe ndị dị ka polyvinyl acetate resin (PVAc), mmanya polyvinyl (PVA), na polyacrylonitrile (PAN) site na polymerization ma ọ bụ copolymerization na monomers ndị ọzọ. A na-eji usoro ndị a eme ihe n'ọtụtụ ebe n'ihe owuwu, akwa akwa, igwe, ọgwụ na ndị na-emezi ala. N'ihi mmepe ngwa ngwa nke ụlọ ọrụ ọnụ na afọ ndị na-adịbeghị anya, mmepụta nke vinyl acetate egosiwo usoro nke na-arịwanye elu kwa afọ, na ngụkọta mmepụta nke vinyl acetate ruru 1970kt na 2018. Ugbu a, n'ihi mmetụta nke ngwaahịa na usoro, ụzọ mmepụta nke vinyl acetate na-agụnye usoro acetylene na usoro ethylene.
1. acetylene usoro
Na 1912, F. Klatte, onye Canada, chọtara vinyl acetate nke mbụ site n'iji ngafe acetylene na acetic acid n'okpuru nrụgide ikuku, na okpomọkụ sitere na 60 ruo 100 ℃, na iji salts mercury mee ihe dị ka ihe na-akpali akpali. Na 1921, German CEI Company mepụtara teknụzụ maka njikọ vapor nke vinyl acetate sitere na acetylene na acetic acid. Kemgbe ahụ, ndị nchọpụta si mba dị iche iche na-anọgide na-eme ka usoro na ọnọdụ maka njikọ nke vinyl acetate si acetylene. Na 1928, Ụlọ ọrụ Hoechst nke Germany guzobere otu 12 kt/a vinyl acetate mmepụta unit, na-aghọta nnukwu mmepụta nke vinyl acetate. Nhazi maka imepụta vinyl acetate site na usoro acetylene bụ nke a:
Isi mmeghachi omume:
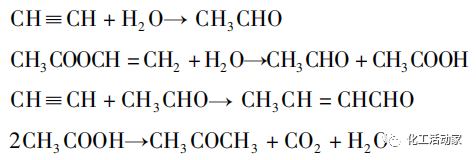
A na-ekewa usoro acetylene n'ime usoro mmiri mmiri na usoro gas.
Ọnọdụ reactant nke usoro mmiri mmiri acetylene bụ mmiri mmiri, na reactor bụ tank mmeghachi omume nwere ngwaọrụ na-akpali akpali. N'ihi adịghị ike nke usoro mmiri mmiri dị ka nhọrọ dị ala na ọtụtụ ngwaahịa, usoro a bụ acetylene gas frasi ugbu a dochie ya.
Dabere na isi mmalite dị iche iche nke nkwadebe acetylene gas, usoro acetylene gas frasi nwere ike kewaa n'ime eke gas acetylene Borden usoro na carbide acetylene Wacker usoro.
Usoro Borden na-eji acetic acid dị ka adsorbent, nke na-eme ka ọnụọgụ nke acetylene dịkwuo mma. Otú ọ dị, ụzọ usoro a siri ike na nkà na ụzụ ma na-achọ ọnụ ahịa dị elu, ya mere, usoro a nwere uru na mpaghara ndị nwere akụ gas.
Usoro Wacker na-eji acetylene na acetic acid emepụtara site na calcium carbide dị ka akụrụngwa, na-eji ihe na-eme ka carbon na-arụ ọrụ dị ka onye na-ebu na zinc acetate dị ka ihe na-arụ ọrụ, na-emepụta Vac n'okpuru nrụgide ikuku na mmeghachi omume okpomọkụ nke 170 ~ 230 ℃. Teknụzụ usoro dị mfe ma nwee ọnụ ahịa mmepụta dị ala, mana enwere adịghị ike dị ka mfu dị mfe nke ihe na-eme ka ihe na-arụ ọrụ, nkwụsi ike na-adịghị mma, oriri ike dị elu, na mmetọ buru ibu.
2. Ethylene usoro
Ethylene, oxygen, na glacial acetic acid bụ ihe atọ eji eme ihe na ethylene njikọ nke vinyl acetate usoro. Akụkụ bụ isi na-arụsi ọrụ ike nke ihe na-akpata bụ nke a na-ahụkarị metal nke asatọ, nke a na-emeghachi n'oge okpomọkụ na nrụgide ụfọdụ. Mgbe nhazi na-esote, a na-enweta vinyl acetate ezubere iche n'ikpeazụ. Nhazi mmeghachi omume bụ nke a:
Isi mmeghachi omume:
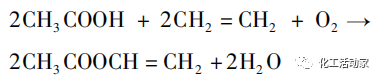
Mmetụta:
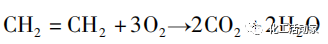
Ụlọ ọrụ Bayer bụ nke mbụ mepụtara usoro ethylene vapor ma tinye ya na mmepụta mmepụta ihe maka mmepụta nke vinyl acetate na 1968. E guzobere ahịrị mmepụta na Hearst na Bayer Corporation na Germany na National Distillers Corporation na United States, n'otu n'otu. Ọ bụ tumadi palladium ma ọ bụ ọla edo na-akwado nkwado acid, dị ka beads silica gel nwere radius nke 4-5mm, yana mgbakwunye nke potassium acetate ụfọdụ, nke nwere ike imeziwanye ọrụ na nhọrọ nke onye na-eme ihe. Usoro maka njikọ nke vinyl acetate site na iji ethylene vapor phase USI usoro yiri usoro Bayer, ma kewara n'ime akụkụ abụọ: njikọ na distillation. Usoro USI nwetara ngwa ọrụ mmepụta ihe na 1969. Ihe ndị na-arụ ọrụ nke onye na-emepụta ihe bụ palladium na platinum, na onye na-enye aka bụ potassium acetate, nke a na-akwado na alumina ụgbọelu. Ọnọdụ mmeghachi omume dị ntakịrị ma ihe na-akpata nwere ogologo ndụ ọrụ, mana mkpụrụ oge ohere dị ala. E jiri ya tụnyere usoro acetylene, usoro ethylene vapor phase na-emewanyewanye nke ọma na nkà na ụzụ, na ihe ndị na-eme ka a na-eji usoro ethylene eme ihe na-aga n'ihu na-arụ ọrụ na nhọrọ nhọrọ. Agbanyeghị, ka ọ dị mkpa ka a nyochagharị kinetics mmeghachi omume na usoro nkwụsị.
Mmepụta nke acetate vinyl site na iji usoro ethylene na-eji ihe nrụpụta akwa akwa tubular jupụtara na ihe mkpali. Ihe oriri gas na-abanye n'ime reactor site n'elu, mgbe ọ na-akpọtụrụ ihe ndina na-akpata, mmeghachi omume catalytic na-eme ka ịmepụta vinyl acetate ezubere iche na obere carbon dioxide sitere na ngwaahịa. N'ihi ọdịdị exothermic nke mmeghachi omume ahụ, a na-ewebata mmiri nrụgide n'ime akụkụ shea nke reactor iji wepụ okpomọkụ mmeghachi omume site na iji vaporization nke mmiri.
E jiri ya tụnyere usoro acetylene, usoro ethylene nwere njirimara nke nhazi ngwaọrụ kọmpat, nnukwu mmepụta, obere ume ike, na mmetọ dị ala, na ọnụ ahịa ngwaahịa ya dị ala karịa nke usoro acetylene. Ogo ngwaahịa ahụ dị elu, na ọnọdụ corrosion adịghị njọ. Ya mere, usoro ethylene ji nwayọọ nwayọọ dochie usoro acetylene mgbe 1970 gachara. Dị ka ọnụ ọgụgụ na-ezughị ezu si kwuo, ihe dị ka 70% nke Vac nke usoro ethylene na-emepụta n'ụwa aghọwo ihe bụ isi nke ụzọ mmepụta Vac.
Ugbu a, teknụzụ mmepụta Vac kachasị elu n'ụwa bụ Usoro Leap nke BP na Celanese's Vantage Process. Tụnyere omenala ofu bed gas adọ ethylene usoro, ndị a abụọ usoro teknụzụ emewo budata mma reactor na catalyst na isi nke unit, na-eme ka aku na uba na nchekwa nke unit arụ ọrụ.
Celanese ewepụtala usoro Vantage ihe akwa ọhụrụ iji gboo nsogbu nke nkesa akwa akwa na-enweghị isi na ntụgharị ethylene dị ala n'otu ụzọ mgbanaka akwa akwa. The reactor e ji mee ihe na usoro a ka bụ ihe ndina edobere, ma ịrịba mma emewo ka mkpali usoro, na ethylene mgbake ngwaọrụ agbakwunyere na ọdụ gas, na-emeri adịghị ike nke omenala edozi bed usoro. Mkpụrụ nke vinyl acetate ngwaahịa dị elu karịa nke ngwaọrụ ndị yiri ya. Ihe na-eme ka usoro a na-eji platinum dị ka isi ihe na-arụ ọrụ, silica gel dị ka onye na-ebu ihe, sodium citrate dị ka onye na-ebelata ego, na ọla ndị ọzọ na-enye aka dị ka lanthanide obere ụwa dị ka praseodymium na neodymium. E jiri ya tụnyere ihe ndị na-akpalite ọdịnala, a na-emeziwanye nhọrọ, ọrụ, na oge ohere nke ihe mkpata.
BP Amoco ewepụtala usoro nhazi gas ethylene nke akwa mmiri, nke a makwaara dị ka usoro Leap Process, ma wuo otu akwa akwa 250 kt/a na Hull, England. Iji usoro a na-emepụta vinyl acetate nwere ike ibelata mmepụta ihe site na 30%, na oge ohere ohere nke onye na-emepụta ihe (1858-2744 g / (L · h-1)) dị elu karịa nke nhazi ihe ndina (700-1200 g / (L · h-1)).
Usoro LeapProcess na-eji reactor akwa akwa na nke mbụ, nke nwere uru ndị a ma e jiri ya tụnyere ihe nrụpụta akwa akwa:
1) N'ime onye na-ahụ maka ihe ndina nke mmiri, ihe na-eme ka ọ na-aga n'ihu na-ejikọta ya n'otu n'otu, si otú ahụ na-enye aka na mgbasa ozi otu nke onye na-akwalite ma na-eme ka otu onye na-akwalite ya na reactor.
2) The fluidized bed reactor nwere ike na-aga n'ihu na-edochie deactivated catalyst na ọhụrụ catalyst n'okpuru ọrụ ọnọdụ.
3) The fluidized bed mmeghachi omume okpomọkụ na-adịgide adịgide, na-ebelata ihe na-akpali akpali n'ihi na-ekpo ọkụ na mpaghara, si otú ịgbatị ndụ ọrụ nke ihe ike.
4) Usoro iwepụ okpomọkụ nke a na-eji na mmiri mmiri na-eme ihe na-eme ka ọ dị mfe nhazi nhazi ma belata olu ya. N'ikwu ya n'ụzọ ọzọ, enwere ike iji otu nrụpụta reactor maka nrụnye kemịkalụ buru ibu, na-eme ka arụmọrụ nke ngwaọrụ ahụ dịkwuo elu.
Oge nzipu: Mar-17-2023